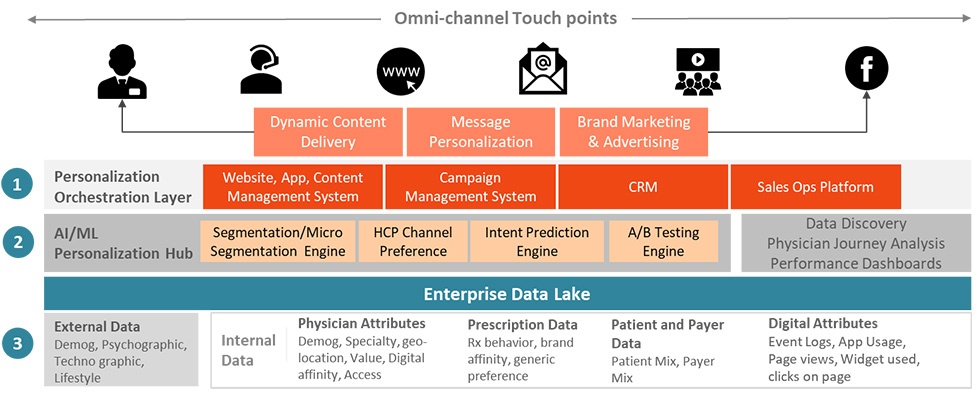
HCP engagement in the new era
The engagement strategies for pharma representatives to connect with HCPs were already in a state of transformation. Covid-19 has only accelerated this process. With fewer HCPs now preferring in-person meetings and with the advent of new technologies, there has been a steady rise in the use of various digital channels like email, social, virtual connects etc. Statistics show that the volume of emails sent to HCPs increased by almost 300% in the last year and the average interaction duration in virtual meetings has increased manifold. All of these developments have compelled the drug companies to reimagine their engagement strategies, maintain a healthy relationship with the HCPs and use the right channels for making the required impact on the HCPs.
Personalization – The need of the hour:
With different technologies at their disposal and HCP preferences differing, pharma companies have realized that they have to change their marketing and engagement tactics to meet the engagement needs of each doctor. Each HCP’s expectation from the interaction is different and the education required for each of them is largely dictated by the patient cohort the HCPs serve. The interest points of HCPs differ and their response to channels differs. So does the response of HCPs to various incentives offered by pharma companies. For example, in one of the recent analyses executed using the Incedo LighthouseTM Platform, we found that the Pediatricians (PD) respond to nutritional rebates much more than their Non-PD counterparts.
Personalization, and sometimes hyper-personalization, therefore is the central theme of customer engagement across domains. HCPs now prefer to be connected to a digital platform of their choice e.g. mobile, email, social, call activity, etc. This behavior may differ across various HCP segments e.g. across therapeutic areas, affiliation, years of experience, and geography, apart from the patient cohorts they serve.
With response data now available to the drug companies, it is possible to derive insights on the HCP preference by various cuts such as segment, sub-segment, geography, etc. The HCP preference and behavior patterns shed light on how receptive they are to digital engagement. This data analysis is leveraged by organizations to analyze the context and content for a digital interaction to derive Next Best Action by answering critical questions related to the message and channel strategies.
Helping Pharmaceuticals understand High Impact Channels using Incedo LighthouseTM
In one of the recent deployments of Incedo LighthouseTM at a pharma organization, the client wanted to understand the most impactful channels for engaging with the HCPs. This was also driven by the CMO agenda to understand the profitable marketing channels to get a bang for the buck. Understanding the segments and sub-segments of HCPs from the visualization and segmentation powered by the Incedo LighthouseTM platform, ML models were built at different therapeutic areas. The marketing investment was translated to input variables specific to the channel and the impact was measured on the overall sales. Using the Data Science workbench of the Incedo LighthouseTM platform, different linear and non-linear ML models were built. The insights from the models were used to derive the contribution of different channels on both the baseline and promotional sales.
Using this, the ROI of channels was determined by various cuts. For example, at a broad level, every marketing dollar spent gave an extra 40 cents as a return. Also, it was understood that among all the channels, digital channels were underinvested and the HCPs responded best to them.
Using the KPI Tree and Cohort Analyzer functionality of the platform, one could see which HCPs were under-reached and had responded well and which were overreached and didn’t respond on certain channels. Using the deep drill-downs, one could go to the affiliation/hospital levels and identify the next action and specific ways to reach them.
Lastly, Incedo LighthouseTM’s advanced visualization capabilities help generate response curves of each channel with further drill-down capabilities. These can be instrumental in simulating the performance of HCP cohorts and channels and identifying the breakeven dollar spend. Laying optimization algorithms on top of it, leveraged from Incedo LighthouseTM’s pre-built accelerators, organizations can get to the channel strategy designed to optimize the HCP engagement through each medium while minimizing the investment.
A US-based, bioanalytical CRO firm was using an ad hoc, manual and static rule-based process to identify and generate leads. It urgently required the transition to an intelligence-led, automated lead scoring engine.
Incedo’s Lighthouse enabled an effective lead scoring system that helped the sales and marketing teams identify which prospects were valuable to the sales funnel and were most likely to convert into paying customers.
This company vastly improved its prospect evaluation and targeting using Incedo Lighthouse’s AI/ML enabled prospecting and dashboard solutions. A significant ~20% more prospects reached the proposal stage and resulted in a robust automated solution for the digital team.
COVID-19 pandemic brought with it a complete disruption to the existing normal operating procedures in most of the industries. The unprecedented situation due to the pandemic has struck some of the business functions disproportionately hard. The most impacted functions in the companies however are those where the workforces relied heavily on “on the field” presence for the execution of their work compared to those functions which could easily be converted into a remote working setup.
From the Life Sciences industry standpoint, the drug promotion via Medical Reps (MR) falls into the prior category. Although the industry as a whole has seen rapid adoption of digital solutions across the workstreams in the ongoing decade, their marketing efforts to the Health Care Providers (HCPs) still heavily rely on the Face to Face (F2F) interaction of the Reps with the Physicians.
This status quo however has been challenged by the ongoing COVID pandemic, with the social-distancing norms in place. There are estimates of 92% drop in F2F HCP engagements in April 2020 compared to 6 months ago[1]. It is also estimated that in the new post-pandemic normal, the frequency of F2F engagements will shift as much as by 65% to quarterly/annual rather than the weekly/monthly norms prevalent pre-COVID2. This is indeed a massive blow to the existing Pharma sales and marketing approach and has seen many of the companies rapidly scale up their digital engagement channels to fill the gap. The use of these digital channels for HCP engagement has seen a 2x increase from their pre-pandemic levels.[2]
The current COVID driven environment has several key implications for the Lifesciences organizations in their effort to meaningfully engage with HCPs.
- Impact on Sales & Marketing Channel Mix – Restrictions on in-person meetings have lead to reduced access to HCPs, canceled/postponed training sessions, and canceled conferences and events, all of which were major marketing methods till now. Pharma and other Lifesciences companies have to accelerate their sluggish digital transformation initiatives and enable a true omnichannel digital experience for HCPs
- Digital engagement channel optimization – The digital omnichannel push needs to account for varying pysician preferences for the type of digital channel engagement, based on factors like therapeutic area, demography, and personal preferences.
- Personalized, contextual messages for better engagement– Physicians at the front lines have to balance innovation and efficiency while dealing with the increased pandemic workload. As a result, engagement and interaction frequency with HCPs have decreased abruptly. With this sudden shift, there is a need for communication to be crisp and contextual for it to be effective.
This brings us to an important question of how the Bio-Pharmaceutical companies should navigate the current shock concerning HCP engagement and what lies ahead for them. Pharma Commercial Teams would need a strategic HCP engagement approach that manages the immediate COVID situation as well as builds capabilities for the new digital-driven normal.
As the Bio-pharma companies scramble to optimize their marketing efforts in the current times, they need to formulate a strategy which tackles the problem in phases:
- Now: Immediate Priorities to manage COVID situation (next 1-3 months) – Set of tactical initiatives and workarounds to the existing HCP engagement methodologies, meant to strictly tackle only the immediate priorities around COVID-19 impact
- Next: Accelerate digital capabilities build-up to drive Omnichannel HCP engagement (in 3-6 months ) – Strategic initiatives to accelerate and deliver a highly engaging digital experience for HCPs. These will fundamentally help in shifting and realignment of biopharma omnichannel engagement capabilities in post-COVID realities.
(Now) Immediate Priorities to manage HCP promotions in COVID situation
As an immediate measure, Bio-Pharma companies need to evaluate the impact of COVID-19 on HCPs’ practice – Rx, patient counts, geographical impact, etc, and Field Reps access to HCPs. It is imperative that Biopharma companies create a COVID control room, which integrates external trigger impact data with internal data sources to truly assess the impact of COVID situation (and potentially other external triggers and shocks) on their sales & marketing plans.
As the COVID impact is quantified, bio-pharma can synthesize the same to adjust the tactical call plans for their promotional activities. The critical parameters to consider while making changes to the call action models would be:
- Incorporate external COVID impact triggers at geo, HCP level
- Defining and quantifying the digital affinity of physicians
- Optimization of cross-channel (Digital & Rep) targeting frequency
- Dynamic adjustments to the call-plan (digital mix, frequency) as the COVID situation evolves
(Next) Accelerate digital capabilities build-up to drive Omnichannel HCP engagement
Once the immediate priorities related to the pandemic are solved, companies can utilize the learnings and key insights from the pandemic times to further advance their digital engagement strategy. The evaluation of what went right and what were the misses in the earlier stage should also be used to formulate a long-term digital and omnichannel engagement strategy. There is also, a lot to learn from Digital-natives who have, highly effectively, leveraged digital channels to driven customer engagement.
Bringing these best-practices from Digital natives together with Bio-pharma context can help accelerate the digital transformation of the industries HCP engagement approach.
| Best-practices and Learnings from Digital Natives | Lifesciences Ecosystem Context |
|---|---|
| Focus on differentiated HCP experience | Physicians have different interaction points, interests, and requirements including clinical content, CMEs, studies, samples, copay coupons, patient counseling material, etc. and hence differentiated experience enables engagement. |
| Volume and variety of data | Pharma has access to multi-dimensional physician data in terms of demography, preferences, prescription patterns, patient/payer mix profiles via claims, digital affinity to micro-segment physicians, and uncover preferences, behaviors, and personalized needs. |
| HCP/Customer Journey management and personalization | Advanced analytics and ML-based approaches can leverage the available data to predict intents, recommend interventions, and seamlessly deliver them via physician engagement platform and processes. |
| Omnichannel execution | Multi-channel interaction provides a foundation platform for delivering these experiences across digital as well as non-digital channels. |
| Measure, Learn & Improve | A/B testing driven digital engagement experimentation anchored on performance-driven, yet responsive targeting strategies. |
To accelerate their digital transformation journey, biopharma companies need to inculcate these best practices into their HCP digital marketing capability. An integrated Digital Engagement solution will help biopharma companies create and deliver omnichannel personalized experiences for HCPs, by enabling real-time AI/ML-driven next-best-action recommendations and precision targeting strategies based on their preference and intents.
COVID pandemic is an unprecedented global event, which will radically alter our behaviors, expectations, and interactions. Earlier rules of engagement are now getting irrelevant at a pace that is faster than ever before. To maintain(and grow) their share of voice and engagement with HCPs, bio-pharma organizations can no longer afford to follow the “digital-addon” approach. They have to fundamentally re-design their HCP engagement framework, as a Digital-driven strategy, $to stay relevant, to stay ahead and keep growing.
The Covid -19 pandemic continues to disrupt the Pharma industry. As uncertainty around the pandemic lingers and refuses to go away, Pharma leaders are facing extraordinary challenges due to the following forces at work:
Rapid shift in demand of drugs due to the impact of Covid-19 – The pandemic has impacted the demand of drugs for various therapeutic areas differently. There is an unprecedented surge in demand for drugs which are being considered as treatment candidates for Covid-19 e.g Remdesivir (Gilead), Actemra (GNE) and Kevzara (Regeneron). There has also been a significant upsurge in demand for symptomatic medicines like antivirals, pain medications and ICU medicines which are used for managing complications from Covid-19. On the other hand, delays in elective surgeries and non-essential treatments have led to huge drop in Rx for many categories, and a rise of product switching in favor of self administered drugs
Geographical risk due to Covid-19 changing very quickly – After weeks of shutdown, some countries and states are cautiously reopening their economy. As regions open up, there are new emerging hotspots which can modify the density of cases, and hence the downstream impact on key decisions for Pharma Cos like inventory allocation for treatment therapies, supplier management and execution of clinical trials. Given the rapidly evolving dynamics with Covid-19, companies need to ensure that they are using the most updated data and case forecasts for decision making
Pharma forecasts are broken – For an Industry which relies very heavily on forecasting, the historic data on which all forecasting, planning and distribution systems are built on has changed. Many of the previous signals used for forecasting like seasonal patterns, events, channel characteristics and patient behavior might not hold true going forward. There are new behaviors like hoarding, preference for self administered drugs and movement to telehealth which challenge pre-existing assumptions. Forecasters need to factor in this “black swan” scenario into their assumptions, and the geographical risk of cases would be one of the key factors impacting Pharma KPIs
Given this scale of disruption, how can Pharma companies solve this?
Unprecedented problems can still be solved with conventional solutions. With the right tools, Data science can provide much needed clarity, direction and guidance on what is happening now , and what is expected to happen ahead. We propose a 5 step approach with a Covid-19 Control Room for Pharma companies which composes of the following components.
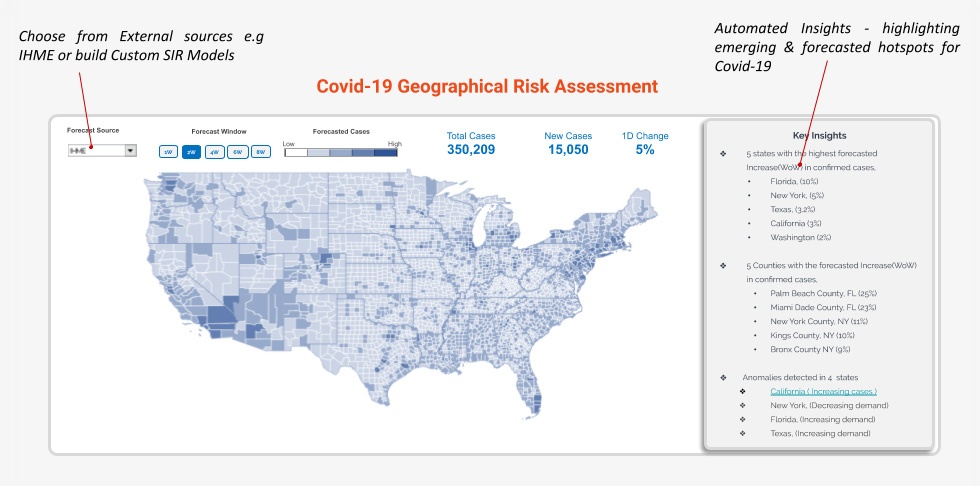
- Covid-19 geographic risk assessment – Assess how the Covid-19 epidemic would play out with estimation of Covid-19 cases at Country, State and County level. There are multiple sources of forecasts like IHME, Northeastern University, Columbia University or you can build custom SIER Models. Models are only as good as their assumptions, so it is advisable to look at forecasts from multiple models to capture the possible range of outcomes for assessment of the geographical risk. A dashboard view like the one below, with the ability to customize the forecast would be a foundation for the Covid-19 Control room. This base estimation of geographical risk can be used to model scenarios for a range of decisions e.g inventory allocation, supplier risk and clinical trial management being some of them

- Segmentation of drugs based on categories of consumption – If you observe the pattern of how demand is getting disrupted across therapeutic areas, there are 3 key demand archetypes that would emerge.
- Direct impact – For drugs which are in late stage clinical trials for treatment of Covid-19 e.g Actemra (GNE), Kevzara (Regeneron), Lopinavir+Ritonavir (AbbVie) – effects of hoarding or upsurge are leading to more than 5X increase in sales, with demand quickly outstripping supply. This has already led to shortages. Remdesivir which received emergency use authorization by FDA might be in shortage for a long time – Gilead reported there’s only enough of it for 200k patients around the world
- Secondary impact – For drugs which help in symptom management e.g pain/anaesthetic drugs like Paracetamol & Ibuprofen, antivirals like Rapivab and respiratory drugs have seen a huge uptick in demand e.g 91% increase in Paracetamol, 27% in Beclometasone, 23% increase in Salbutamol in the first week of Mar’20 compared to Mar’19. There are also a class of drugs used for Covid-19 complication management e.g ICU drugs like Epinephrine, Fentanyl and Oxycodone which have seen demand surges and reported shortages
- Negative or no impact – For some categories, demand has fallen sharply. In office administration volumes show huge drop in demand including certain categories of prescription drugs e.g -45% Rx for Pediatric Antibiotics. There are shifts based on mode of administration e.g IV administered oncology therapies show decreased demand relative to oral therapies
- Calibrate demand sensing for each demand archetype – Understanding the demand archetype of drugs in the company’s portfolio would enable forecasters to calibrate the demand post disruption and improve the accuracy of their forecasts. One of the key signals of drug demand at the distributor level is the geographic risk. For some like Actemra and Kevzara, the increase in demand would be directly proportional to the number of cases with Covid-19 in any region. E.g we know that Kevzara is a treatment candidate for patients with severe pneumonia due to Covid-19. Signals like number of expected cases, admission rates, patient demographics & access to the drug can be used to derive an accurate estimate of the demand for the drug. Similar analysis is needed based on demand archetype to calibrate forecasting techniques for other therapeutic areas given this ‘structural break’ in historic time series data due to Covid-19
- Develop strategies for short, medium and long term – Once you have a sense of impact on the therapeutic area, organize your efforts for the Covid-19 Control room which would provide a perspective on short term ‘Crisis management’, medium term ‘Risk management’ and long term ‘Restoration to normal’ initiatives. For Supply chains, the initiatives would be:
- Short term Crisis Management – Inventory risk assessment & allocation
- Medium term Risk management – exploring options for ramping up production, managing supplier risk and reducing lead time
- Long term Restoration to Normal – capacity planning, planning for recurring cycles of pandemic
For example, here is an illustration of Inventory Risk assessment with key insights on Inventory Allocation at county level for a demand archetype with direct Covid impact. An inventory risk assessment and allocation solution would compute the mismatch between demand and supply – measured by the ‘shortfall from required DoH’ to surface alerts. This would ensure that inventory allocation is optimal – precious drugs are sent to the critical locations and hospitals who need it most.
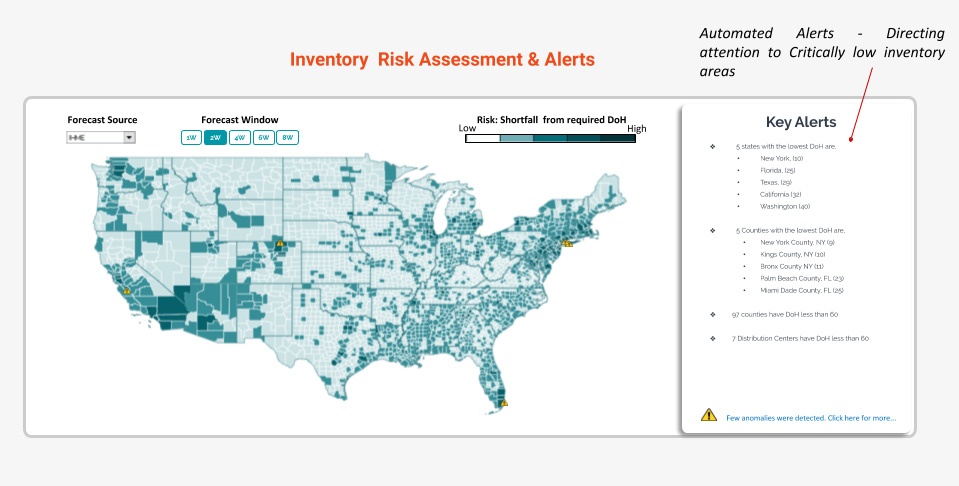
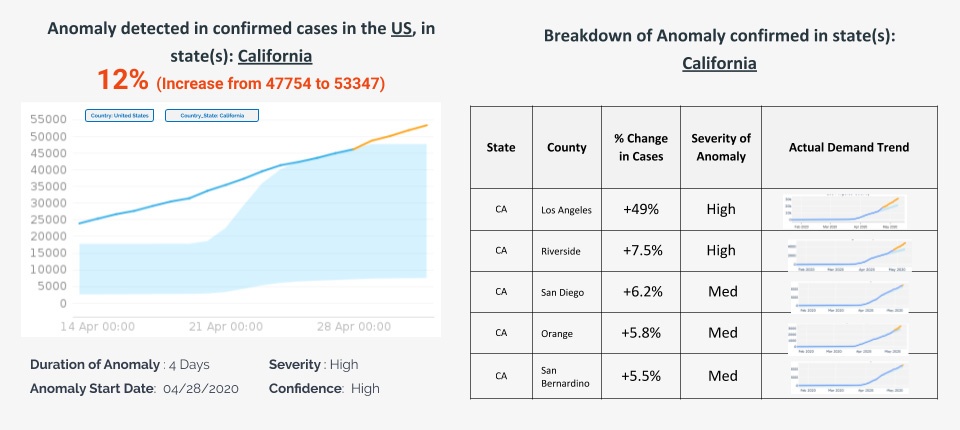
- Build early warning indicators – Covid-19 infections, as some expect, might stay and relapse long into the future – even after the first wave. Machine learning can be used to constantly analyze and correlate parameters like case rate, death rate and growths with anomaly detection systems which detect shifts in cases and identify emerging hotspots of infection. This will help companies quickly recalibrate decisions e.g here is a view of how an autonomous Anomaly detection system for the Covid Control room is built at country, state and county levels enabling decision makers to zoom in and out to identify hotspots quickly.
As we have all experienced, every day is a new unprecedented chapter in this outbreak of Covid-19. Strategies leveraging data and tools at our disposal can help Pharma companies win the battle against this pandemic. Companies that execute on these strategies will have a clearer view of what is expected to happen, and hence better prepared to face the challenges which lie ahead.
This Article is part 2 in the series – ‘Managing Pharma Supply Chains in times of Covid-19‘
For more insights on how Pharma companies can Optimize their Supply chains, please click here.
FDA Guidelines on Real World Data and Real-World Evidence
“As the breadth and reliability of RWE increases, so do the opportunities for FDA to make use of this information”, noted Scott Gottlieb, former FDA Commissioner National Academies of Science, Engineering, and Medicine while examining the impact of RWE on medical product development.
FDA has a long history of using RWE to monitor and evaluate the safety of drug products after they are approved. Real world data traditionally come from a variety of sources such as data derived from electronic health records (EHRs), medical claims and billing data, data from product and disease registries, patient-generated data, including from in-home-use settings, and even data from mobile devices that can inform on health status. With such ever-increasing reliability of real world data, FDA published its Framework for FDA’s Real World Evidence program. It laid out a framework for evaluating the potential use of real-world evidence (RWE) to help support the approval of a new indication for a drug already approved under section 505(c) of the FD&C Act or to help support or satisfy drug post-approval study requirements.
Further, the May 2019 draft guidelines, “Submitting Documents Using Real-World Data (RWD) and Real-World Evidence (RWE) to FDA for Drugs and Biologics,” encourage sponsors using RWD to generate RWE as part of a regulatory submission for investigational new drug applications (INDs), new drug applications (NDAs) and biologics license applications (BLAs).
These developments are growing acknowledgement by FDA on the need for Real-World Evidence across RCTs, single arm trials and observational studies, to enhance clinical research and support regulatory decision making.
Are Pharma and CROs ready ?
FDA has made RWD and RWE a “top strategic priority”. Medical Research is at a turning point – with an abundance of real-world data from a wide variety of sources ranging from EHRs to wearables like Fitbits delivering terabytes of data, healthcare practitioners are pushing ahead on delivering personalized care, while acknowledgingthe value of real world data and evidence.
It’s a redefining moment for Lifesciences industry – the professionals across the industry understand that RWD will transform not only clinical development process but also commercialization and reimbursement decisions. However, Life Sciences – Pharma, Biotechs, CROs – are yet to widely adopt RWD insights in an institutional way because many are unsure of the road forward and are facing several roadblocks.
There are challenges such as:
- Inconsistent RWE data collection and quality while increasing complications due to new data sources, resulting in data fragmentation.
In Lifesciences industry having data has never been a problem, however, the ability to stitch together a robust patient-journey data has been complicated. Once the patient health journey data is complete, it allows researchers to compare interventions and outcomes more meaningfully. With this the objective to fully understand the impact of different clinical options can be fulfilled. More importantly, the insights give researchers an ability to understand patient’s challenges and assess how their medical products perform when the patient need is the highest.
RWD researchers today typically use Electronic Health Record, Health Insurance Claims and Population Health sources of aggregated data for their research. There is a growing desire to leverage several new sources of patient data to make evidence generation more robust, insightful, and richer. Genomic data, biomarkers and digital data i.e. data generated from mobile devices, wearables, health apps or other biometric devices – are topmost in the priority order across organizations and researchers for real world insight generation.
However, organizations usually encounter challenges in stitching together the patient journey across these varied RWD data sets, due to their inherent nuances and complications.
- Lack of robust internal resources with the needed RWD experience and expertise.
The current nature of RWD initiatives or studies is very bespoke within lifesciences organizations. The typical approach has been to execute each study in a silo, usually dependent upon a few researchers and biostatisticians. Such an approach has led to significant gap in terms of demand for pre and post commercialization real world analytics, and the capabilities of the organization to fulfill the demand.
With hurdles such as lack of limited RWE skilled resource pool, poor knowledge sharing, lack of standard methodologies and next to no reusability, this gap continues to widen.
- Need for technology to manage and analyze data and provide RWE.
With the exponentially growing complexity and volume of different emerging RWD data sources,Lifescience and Pharma organization are seen to be insufficient for analysis and evidence generation. For example, with the increasing use of wearables and biometric devices, digitally patient reported data is becoming mainstream for clinical drug trials, while the traditional data platforms are struggling to process this real-time, streaming source of patient health information. Similarly, intelligent insight generation from EHR data is something which is beyond capabilities of current systems, as it requires new-age AI and NLP capabilities to process text data which is missing in traditional systems.
Advanced data and AI/ML driven computing technologies are critical to aggregate consistent and robust patient-journey data from these RWD sources.
Strategy for institutionalizing Real-World Data and Evidence Generation
The role of real world evidence in drug development and post-commercialization evidence monitoring is really turning the corner. So far, only payers were demanding evidence as part of the access and reimbursement process; but with FDA putting its full weight behind acknowledging it as a critical part of its regulatory framework, it right time for Pharma and CROs to build their real world data capabilities at the institutional level.
Evolving RWE as a core institutional capability and to give it necessary organizational focus, Pharma and CROs has to invest in and build out centralized “RWE center of excellence”, with a federated ecosystem of evidence generation.
- The RWE center of excellence must focus on developing core capabilities around RWE data acquisitions, standards, technology and processes.
- Develops and enforces RWE data collection and quality standards
- Acquires, develops best-in-class Big data (high complexity, structured, unstructured, streaming) processing and management capabilities for enterprise RWE data management
- Design and implements standard analytics methodologies to ensure consistency in quality of evidence generated across the organization
- Institutionalizes RWE knowledge base via structured training and knowledge management, supporting the rapid ramp-up of enterprise RWE skilled resources
- Develops standards and SOPs across RWE processes and evidence reporting
- Drives execution of strategic and highly critical evidence studies
- The broader federated ecosystem – regional, therapeutic area, product line-based teams – focus on study design and execution as well as developing partnerships and alliances.
- Execute critical RWE studies for their respective regions, therapeutic areas or product portfolios
- Drives local, regional or therapeutics areas partnerships with RWE data owners, peers, providers and other health industry stakeholders
Such a “core” Center of Excellence integrated with “federated” evidence generation strategy, is critical to bring the required scale to overall RWE generation and enablement by marrying COE driven standardization with the flexibility to meet growing specific RWE needs across the organization.
Even in stable times, Pharma supply chains are fragile and as complex as they can get. As the Covid-19 pandemic continues to wreak havoc on countries around the world, Pharmaceutical supply chains have come under immense pressure. In this article, we would cover some of the key challenges which Pharma supply chain Executives face on the frontlines, and how Analytics and Data Science can be leveraged to overcome these challenges.
Covid-19 disruptions are going to test the strength of Pharma Supply Chains:
- Stockouts are a real risk: According to a study by University of Minnesota, 80% of the drugs marketed in the United States, including 19 of the 20 top-selling brand names, are made overseas. The global nature of supply chains, regulatory challenges and high uncertainty makes stockouts a real risk – jeopardizing the health of millions of patients who depend on life saving drugs
- Operational metrics for Pharma Cos would need drastic improvement: With an average inventory of 258 DoH , the Pharma industry has one of the largest inventory stockpiles – 2-4X larger than FMCG at 72 DoH. However, given the staggering scale of this pandemic and the real risk of stockouts, Pharmas would need to rethink and optimize their inventory allocation strategies to ensure that the current inventory of drugs is allocated to the channels and regions with the most urgent need
- Shift in Consumption Patterns: As drugs are identified as potential treatments for Covid-19, the demand may quickly surpass the supply. These drastic shifts in consumption patterns have already been observed with Covid-19 treatment candidates e.g Genentech’s Actemra, Sanofi & Regeneron‘s Kevzara and Gilead’s Remdesivir, an experimental drug for Covid-19. In early April, the FDA reported shortages of hydroxychloroquine and chloroquine, antimalarial drugs that were speculated to be front-runners for a possible Covid-19 therapeutic. This shortage has impacted patients with Lupus, where chloroquine is a life saving drug
- Long-term Cyclicity of a Recurring Pandemic: Taking a page from history, the Spanish Flu epidemic hit in waves, the second wave more lethal than the first. If the Covid-19 virus proves to be seasonal, the impact of the pandemic might happen in waves over a 1-3 year period before stabilizing. Pharma Cos should be prepared for detecting and responding to new drivers of demand with very high momentum. Some new drivers of demand could be – preference for self administered drugs due to a drop in hospital visits, higher propensity for hoarding and increased demand for normal uses of certain drugs e.g acetaminophen to treat fever & flu symptoms
Given the scale of disruption, how can Pharma Supply Chain Executives approach these challenges ?
A combination of strategic and operational moves leveraging Analytics and Data Science capabilities will help you get to the critical insights necessary for getting started.
- Gain a realistic view of your current state: Creating a transparent view of your supply chain and assessing the current state is your first step. Quick dashboards and ad-hoc analysis will give you a perspective of what is happening on the ground, and in the moment. The views should be built to assess 3 key stages in your Supply chain:
- Multi-tier Supply Assessment– What are the most critical components of Supply? What is the risk of interruption? What is the next best action for high risk Suppliers?
- Inventory Audit– Where does your allocated Inventory lie both in-house and with distributors? What amount of this Inventory is finished goods vs blocked for quality control and testing? What is the volume of Inventory in transit?
- Demand – What is the most realistic estimate of customer demand? Are there any specific NDCs with disproportionate impact on demand? How is the demand distributed at the Distributor, Geo and NDC level? Are the underlying assumptions of demand signals still robust? What are the emerging drivers of demand which might be getting missed?
- Break down your perspective – Short term and Medium term: Organize your efforts with a Covid-19 Command center which would provide a perspective on ‘Short term Crisis management’ and ‘Medium term Planning Ahead’ initiatives. This would ensure that teams on the ground continue to have bias for action, without getting blindsided by what’s coming ahead.
- Crisis Management teams– Focus on the most immediate tasks where speed is of essence. This team would focus on the most high impact disruptions and build quick dashboards/reports to get a transparent view of the current situation and generate critical insights for operational teams on the field
- Planning Ahead teams– Look ahead to answer questions on mid-term and long term impact – like testing underlying assumptions, bringing in new intelligence from external analysis, identifying and integrating new signals and data sources into the analysis and developing scenarios for the future
- Develop Scenarios for multiple versions of the future: The nature of the current Covid-19 pandemic is such that the arc of impact would be varied and staggered across the world. Take the US for example, every state and county is experiencing the pandemic differently. Hence, supply chain teams need to develop a scenario based decision making frame to assess how the pandemic would pan out, and what are their best moves at the moment . Here , the scenarios need to be built at 2 levels :
- External Scenarios– Evaluate impact of the pandemic and effectiveness of the response at macro and micro levels across countries, states and down to county levels on key metrics like demand and supply
- Internal Scenarios – Simulate impact of moves based on a Pharma’s ability to respond to the crisis. This would involve tweaking Supply chain parameters like manufacturing and shipping lead times, safety stock assumptions to identify what is the next best action that should be taken by Channel Inventory, Demand Planning and Manufacturing teams in the medium and long term
- Factor for Uncertainty and Anomalies: During times of uncertainty , one of the most powerful tools in the arsenal of Data Science is Anomaly detection. Over here, the unknowns are shifts in consumption patterns and cyclicity of recurring pandemic which would be hard to detect with human judgement. Once fed with the historic data, powerful ML algorithms can help you quickly spot unknown unknowns in your data, down to the most granular levels of detail – helping you set up algorithmic trigger points to flag alerts. This should be one of the key pillars of response for the ‘Medium term Planning Ahead’ workstream. Some examples of metrics to be looked at:
- Analyzing past demand patterns with anomaly detection models would help quickly spot which NDCs are impacted by the shifts in consumption patterns to predict stock outs at zip code level
- Anomaly detection algorithms to flag emerging new hotspots of emerging Covid-19 cases at a country, state and county level impacting distribution and logistics
- Be prepared for a fundamental change in the nature of your Data: With more than 3 billion people in lockdown, this epidemic will bring dramatic changes in patient, distributor and regulatory behaviors around the world. Covid-19 is a perfect example of a ‘structural break’ in your time series data – with implications both in the short and the long term. Pharma companies might need to look for unconventional sources of data for getting insights during times of uncertainty. For example – Google Search trends have been found to be good predictors of demand for certain types of drugs, and can also help us find emerging Covid-19 outbreaks. They can also reveal symptoms like ‘loss of smell’ that at first went undetected.
The current crisis has plunged entire countries and the Pharma industry into times of uncertainty. Building a transparent view of the current state, scenario based planning and proactive detection of anomalies are key tools on the frontlines for defense.
By acting intentionally today and using the tools at our disposal, Pharma companies can weather this crisis, emerging stronger and building resilience for the future. And in the process, enhancing and saving many lives around the world.
Patient Marketing: Changing Landscape
I have seen patient marketing in HealthCare undergo significant change in a rapidly transforming commercial and digital environment. There are numerous factors disrupting the status quo:
- Increasing role of patients in treatment and care
- Competition for “high-value” patients for “elective” provider service lines (e.g. Knee Replacement)
- The availability of large “volume” and “variety” of data, and tools to organize and mine it
- Growing importance of alternate channels (increasingly digital) to reach prospective patients
- A steady increase in HealthCare digital marketing spend; albeit it’s still significantly lower compared to other industries
Personalization: From Consumer Internet to Patient Acquisition Marketing
Traditional patient marketing by HealthCare providers presents a number of challenges:
- It is rules driven broad based targeting and does not mine available data to create a patient 360 profile for personalized outreach
- The importance of patient journey map and the role of “influencers” is not explicitly mapped
- Channel preference is not explicitly modelled or accounted for
- Past campaigns and channel outreach not incorporated
Given the changes in the commercial marketing environment, it is imperative that HealthCare Providers drive personalization in patient targeting – the “right patients” to target with the “right channel outreach” and the “right message”
The native digital firms are at the cutting edge of using advanced AI/ML modelling techniques to drive effective consumer targeting. National retailers’ online loyalty programs are a good example of how these firms leverage data to optimize targeting. They use multi-year history of data (website visits, clicks and conversions along with email opens, clicks and subsequent conversions) in Bayesian machine learning models, to create a propensity score for each product-consumer segment. This is employed for personalized e-marketing and user experience, with estimated marketing ROI improvement of around 20 percentage points.
Some key learnings from these firms include:
- Untargeted outreach is ineffective – users tune them out
- Machine learning models can leverage history of user behaviour on emails, apps, website and offline data such as phone calls or sales contacts
- The models can be operationalized to power personalized experience on all “user touch points”, such as emails, website, call centre etc.
- Personalized experiences leads to user delight and increased response rates, as well as reduce churn and improve ROI of marketing channel promotions.
Bringing Learnings and Best Practices from Digital Native Firms to HealthCare
In order to leveraging the experience from digital native firms, it is imperative that we bring the “Best Practices and Learnings” to the HealthCare business context:
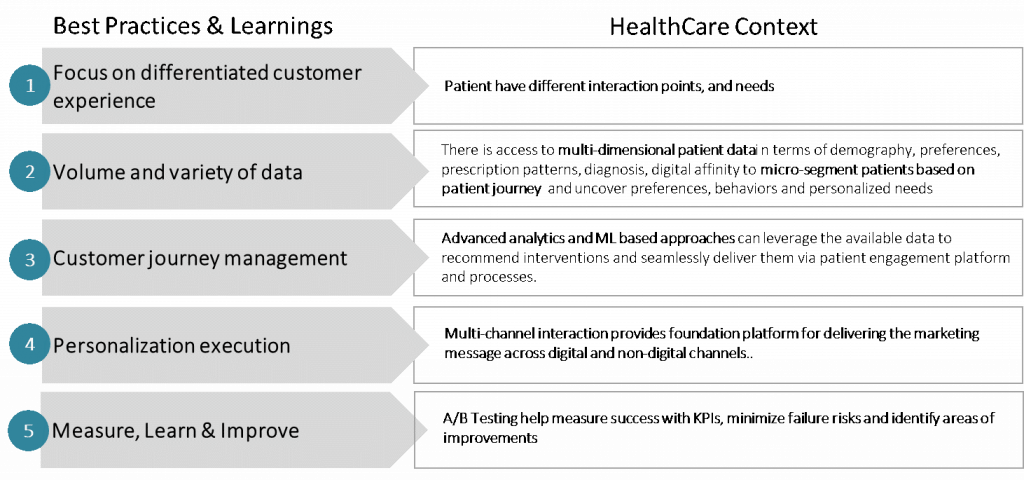
Patient Acquisition Marketing Personalization: Problem-Solving Framework
A structured problem solving approach is necessary to analyse personalization in patient marketing. A key part of this framework is to link the
propensity prediction and recommendation to patient segmentation and available data (patients with some history within our system vs. new prospects).
In addition, the prioritization must be linked to core business objectives e.g. drive business in “orthopaedic surgery line of service as part of overall expansion and profitability strategy”
A broad outline of the problem solving framework and its constituent parts is shown in the illustration below:
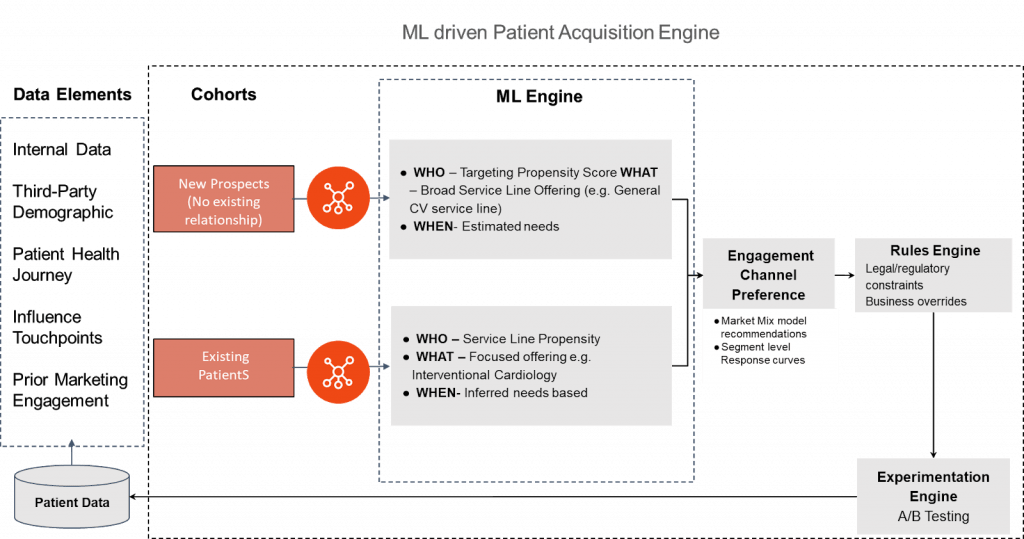
In my experience, the key to driving personalization in patient acquisition is:
- Creating a 360 patient profile including the journey map for existing patients
- Choice of “appropriate” ML modelling approach based on patient segment (existing vs. new prospect)
- Channel preference modelling based on available data and/or experimentation approach to try out alternative outreach strategies
- Ability to augment modelling results with business priority driven rules
- Integration of existing patient outreach infrastructure to deliver marketing message via the appropriate channel
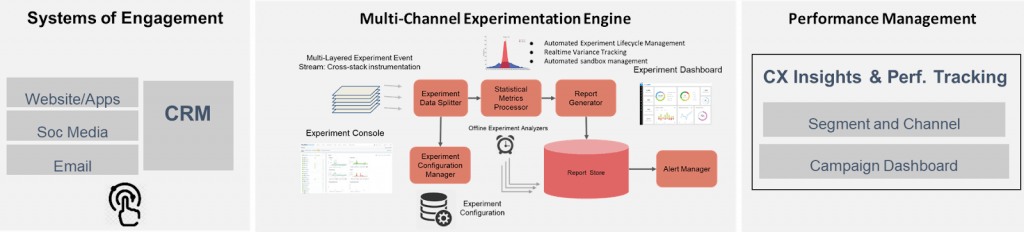
Program Execution
There is great diversity across organizations. In order to successfully execute personalized targeting and digital marketing solutions, it is also imperative to define the right operating model that aligns with business objectives, organizational strengths, and the right partner(s).
Here is a brief outline of an execution approach that I have been seen work well:
- The output from the “personalization” analysis is executed thru the existing patient engagement platforms
- It is also imperative to deploy an A/B testing framework to improve our channel outreach and messaging over time
- Finally, a “system of insights” that can track the effectiveness of our marketing efforts and deliver both “insights” and “course-correction” recommendations is key for ongoing success and to optimize program ROI
In conclusion, a data-driven approach that leverages the best in class AI/ML modelling; implemented within the context of our organizational engagement model, and focused on key business metrics is critical to driving successful implementation of patient marketing programs.